They are multipotent, interact closely with endothelial cells, and are involved in immunosurveillance: the heterogeneous group of pericytes. These cells build the wall of blood vessels, and play a crucial role in the formation and maintenance of the vasculature. Due to their involvement in various pathophysiologies such as tumor angiogenesis, scientists are beginning to exploit their potential for therapeutic approaches.
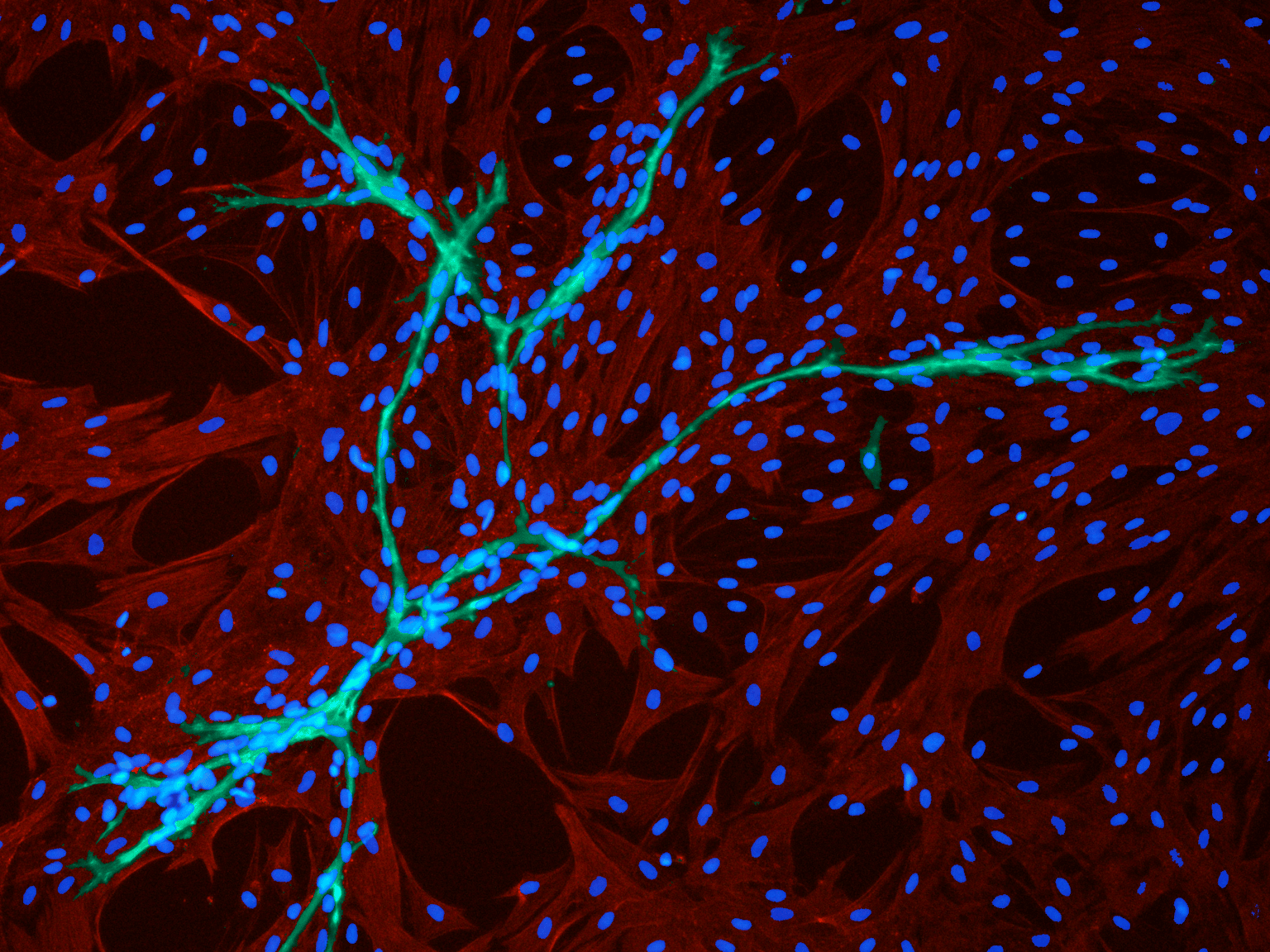
Interacting with endothelial cells
Interacting with endothelial cells: Immunofluorescence staining of cell cultures in EGM2 medium from PromoCell shows pericytes (green, positive for the expression of α-smooth muscle actin) and surrounding endothelial cells (red, CD31 positive). Picture kindly provided by Dr. Frédéric Deschaseaux.)
More than 140 years ago, Charles Marie Benjamin Rouget, a French physiologist, discovered pericytes, contractile cells that build a spider web around small vessels (Herndon et al., 2017). They are present in every vascularized tissue of the body, covering between 22 percent and 99 percent of the endothelial cell surface. Although scientists have known about pericytes for many years, they were not really sure of the role of these cells until recently. Even now, fundamental questions about where pericytes come from, what they do, and what they can become remain unanswered. However, new insights about cell surface markers, about interactions between pericytes and other cell types, and about their role in diseases are lifting the veil from their world (Bergers and Song, 2005).
A particular challenge when working with pericytes is their heterogeneity. They display high variability in terms of ontogeny, localization and morphology. Moreover, some of them have stem cell properties, and can differentiate into adipocytes, osteoblasts, smooth muscle cells, chondroblasts and neural-like cells (Bouacida et al., 2012). In the past, many studies have focused on endothelial cells, and only in recent years has the importance of pericytes for the integrity of the vasculature been discovered. Pericytes are essential for maintaining a healthy vasculature. They work together with endothelial cells, building a functional unit and influencing each other in regulating microvascular stability, both through direct contact and through signaling molecules (Kelly-Goss et al., 2014). Pericytes also play important roles in regulating capillary blood flow, in angiogenesis, in hematopoiesis, in maintaining the permeability of the blood-brain barrier, in wound healing, and in leukocyte trafficking (Armulik et al., 2011).

Dr. Frédéric Deschaseaux, at the STROMALab in Toulouse, France, is studying pericytes, with the aim of developing tissue engineering and regenerative medicine protocols. Picture kindly provided by Dr. Frédéric Deschaseaux.
Definition of pericytes – a heterogeneous population of stem cells
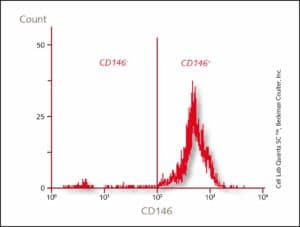
Expression of CD146
Flow cytometric analysis of PromoCell Pericytes show that more than 98 percent of the cells are CD146+.
Dysfunctions lead to major pathological processes: pericytes in cancer
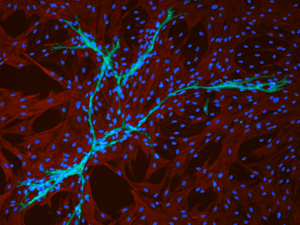
Interacting with endothelial cells
Interacting with endothelial cells: Immunofluorescence staining of cell cultures in EGM2 medium from PromoCell shows pericytes (green, positive for the expression of α-smooth muscle actin) and surrounding endothelial cells (red, CD31 positive). Picture kindly provided by Dr. Frédéric Deschaseaux.)
A new tool in regenerative medicine
There is another side to pericytes. They are very important during tissue development, and in tissue regeneration processes, as they can be activated in response to tissue injury to contribute to the healing processes (van Dijk et al, 2015). “Pericytes play a strong role during tissue formation. When endothelial cells migrate into newly formed tissues during angiogenesis, pericyte populations lead the way to the top of the new vessels. These pericytes can then differentiate into mature cells of different specificities. When looking at bone injuries, or during bone development, vessels are formed by new cells in close interaction with pericytes, which express osteoblastic features,” explains Deschaseaux (Ono et al., 2014). The regenerative potential of pericytes depends on the tissue they come from, and this characteristic has to be taken into account when designing pericyte-based regenerative strategies (Herrmann et al., 2016). “Pericytes show strong differentiation and proliferation potential in vitro. Both properties are very useful for cell therapy approaches. In addition, other perivascular cell types, adventitial cells, are described as “stem-like cells” exhibiting strong overlap on phenotype with pericytes (Hu and Xu, 2011). Therefore, efforts should continue that will enable scientists to clearly distinguish all different perivascular cells. New technologies such as single-cell RNA sequencing can provide data to better typify these cells, which will allow the development of highly precise regenerative therapies,” concludes Deschaseaux.Differences and Similarities Between Pericyte Progenitors and MSCs
Dr. Deschaseaux analyzed with a group of researchers how culture conditions influence cell behavior of mesenchymal stem cells and pericyte progenitors. They assessed surface markers after cultivating the cells in MSC medium and endothelial cell growth medium. Read their paper and find out more about their results and what this means for the engraftment potential of those cells.
read the paper now


